
Under infection conditions, NLRs initiate the inflammatory signaling through the recognition of pathogen associated molecular patterns from viruses, parasites or pathogenic bacteria. It can be either beneficial to assist the clearance of the pathogen, or detrimental to exacerbate the symptoms due to excessive inflammation. Our goal is to use structural biology and biophysics to study the signaling processes that fine-tune the inflammatory response.

Viral infection may produce Immune Booster to be recognized by PRRs to activate innate immune signaling. In the meantime, it may also produce immune silencer to hijack the host factors in order to escape from the immune surveillance. Our goal is to use structural biology to depict the molecular details of the host-virulence factor interaction, further guiding the drug discovery targeting infectious diseases.
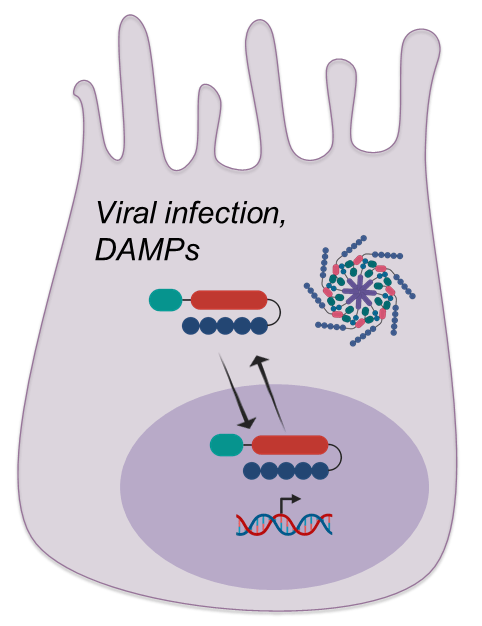
The primary function of NLRs is to form inflammasome complex in cytosol to induce cytokine secretion and inflammatory cell death. The functions of NLRs in different subcellular locations are less studied. Our goal is to draw connections between infection events and chronic diseases by studying the subcellular translocation of NLRs and their inflammasome-independent functions with structural biology and biochemistry approach.